Semiconductors can be classified according to their position in the periodic table, and this classification helps in understanding their properties and applications.
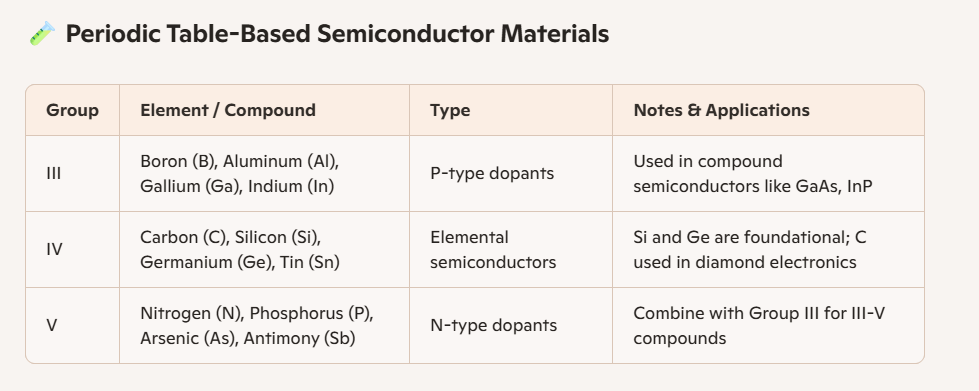
Group IV elements such as silicon, germanium, and tin are elemental semiconductors, with silicon being the most widely used due to its stability and abundance.
Group VI elements like selenium and tellurium also exhibit semiconducting behavior, though they are less common in mainstream electronics.
Compound semiconductors are formed by combining elements from different groups, such as III–V compounds like gallium arsenide (GaAs) and indium phosphide (InP), which are prized for their high electron mobility and use in optoelectronics.
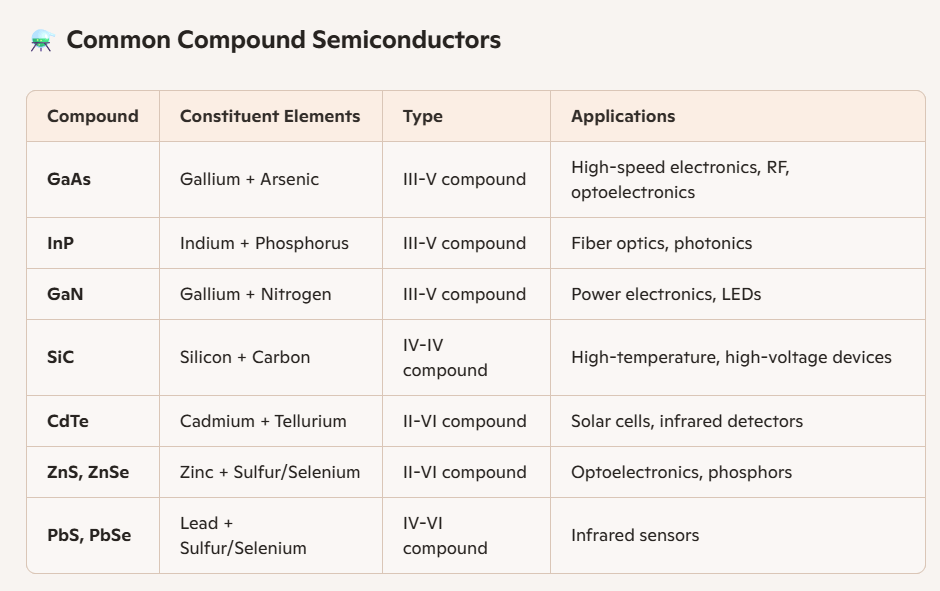
II–VI compounds such as zinc oxide (ZnO) and cadmium sulfide (CdS) are often used in photodetectors and solar cells.

Each category offers unique advantages, from the cost-effectiveness of silicon to the high-speed performance of GaAs.
The periodic table of semiconductors thus provides a roadmap for engineers and scientists to select the right material for specific applications, ensuring progress in electronics, communications, and renewable energy technologies.
BitcoinVersus.Tech Editor’s Note:
We volunteer daily to ensure the credibility of the information on this platform is Verifiably True.
If you would like to support to help further secure the integrity of our research initiatives, please donate here: bc1qrved9tfquym6u3age7xhmnkjs2lq8j9aulperagkuhtuk5w5c35ssfpge8
BitcoinVersus.tech is not a financial advisor. This media platform reports on financial subjects purely for informational purposes.

Leave a comment